NOTE: Yamasa SP-D Kit EIA II has been discontinued by the manufacturer.
Description
The SP-D Kit EIA II, developed and manufactured in Japan by Yamasa Corporation, is a quantitative immunoassay for determination of human surfactant protein D (SP-D) in serum.
SP-D is a 43-kDa protein with a collagen-like domain. Its four trimers assemble in vivo to form a dodecamer. SP-D is known as a lung-specific protein because it is mainly produced by alveolar type II epithelial and club cells.
SP-D plays important roles in the biological defense mechanism in the airway-alveolar system. For example, it promotes bacterial phagocytosis by alveolar macrophages. On the other hand, SP-D in the alveoli is transferred to the blood and increased in several types of interstitial pneumonia, such as idiopathic pulmonary fibrosis, collagen disease lung, and drug-induced lung injury.
Type
Assay principle of this kit is based on the solid phase enzyme-linked immunosorbent assay (ELISA).
Other names
Pulmonary surfactant-associated protein D, SFTPD, PSP-D, Collectin-7, Lung surfactant protein D, SFTP4
Principle of Method
This kit is intended for measuring serum SP-D by enzyme immunoassay (EIA) using the microplate solid-phase method. A sample is added to a microplate with an immobilized mouse anti-human SP-D monoclonal antibody. Then, it is reacted with a horseradish peroxidase-labeled mouse anti-human SP-D monoclonal antibody with a recognition site different from that of the solid-phase antibody. In this process, a solid-phase antibody-antigen (SP-D)-labeled antibody complex is formed. Subsequently, the amount of SP-D in the sample is quantified from absorbance at 450 nm in the enzyme-substrate reaction (color reaction).

Sample Types
Serum
Sample Volume
Serum: 9.09 µL (actual amount of sample loaded in well after dilution)
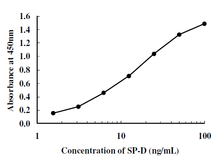
Standard Curve

Calibration Range: 1.56-100 ng/mL
Usage
This product is intended for laboratory research use only. Not for use in diagnostic procedures.
Resources
Want to learn more about SP-D? We have compiled the links below which contain information that you may find interesting:
- UniProt Knowledgebase: UniProtKB - P35247 (SFTPD_HUMAN): Pulmonary surfactant-associated protein D
- PubChem Gene Summary: SFTPD - surfactant protein D (human)
- Crouch, E C. “Surfactant protein-D and pulmonary host defense.” Respiratory research vol. 1,2 (2000): 93-108. doi:10.1186/rr19
- Sorensen, Grith L. “Surfactant Protein D in Respiratory and Non-Respiratory Diseases.” Frontiers in medicine vol. 5 18. 8 Feb. 2018, doi:10.3389/fmed.2018.00018
- Hsieh, Miao-Hsi et al. “Human Surfactant Protein D Binds Spike Protein and Acts as an Entry Inhibitor of SARS-CoV-2 Pseudotyped Viral Particles.” Frontiers in immunology vol. 12 641360. 14 May. 2021, doi:10.3389/fimmu.2021.641360
- Waseda, Yuko et al. “Surfactant protein D: a useful marker for differentiation of drug-induced pneumonia and bacterial pneumonia.” Pneumonia (Nathan Qld.) vol. 13,1 11. 5 Jun. 2021, doi:10.1186/s41479-021-00087-6
References to Summary
- Persson A et al. (1988). Biochemistry. 27. 8576.
- Nagae H et al. (1996). Jpn J Med Pharm Sci. 36. 803-808.
- Honda Y et al. (1996). Jpn J Med Pharm Sci. 36. 809-815.
- Tanaka M et al. (2008). Jpn J Med Pharm Sci. 59. 439-446.
RELATED PRODUCTS
Ilex Life Sciences LLC is an authorized distributor of Yamasa SP-D Kit EIA II.